Understanding the Chemistry Behind Softer, Cleaner Water for Your Home
Key Takeaways
- Protect your plumbing and appliances by understanding how ion exchange replaces damaging minerals with harmless sodium or potassium ions.
- Master the mechanics of resin beads and how they act like magnets to trap scale-causing contaminants.
- Keep your system efficient with our simple guide to regeneration cycles and maintenance schedules.
Dealing with crusty white scale on your faucets, spots on your glassware, and dry, itchy skin can be incredibly frustrating for any homeowner. These are the classic signs of hard water, and while the solution often involves an “ion exchange” system, the term itself can sound like a daunting chemistry experiment. The good news is that the process is actually quite straightforward and incredibly effective at protecting your plumbing and appliances. We’re here to demystify the science behind this technology so you can understand exactly how it improves your home’s water quality and saves you money in the long run.
What Is an Ion Exchange Water System?

At its core, an ion exchange water system is a physical and chemical process used to remove specific impurities from your water supply. You will most commonly find this technology inside a standard home water softener. Before diving into the general process, it is helpful to recognize the three primary system components that make this technology work:
- Mineral Tank: The central chamber where the actual water softening and ion exchange chemical reaction happens.
- Brine Tank: A secondary storage container that holds the high-concentration salt (or potassium) solution required to clean the system.
- Control Valve: The brain of the operation, sitting on top of the mineral tank to meter water usage and automatically trigger the regeneration cycles.
The primary goal of this setup is “softening” or “demineralization,” which involves removing dissolved solids like calcium and magnesium that make water hard. Unlike simple mechanical filters that trap debris in a physical mesh, an ion exchange water filter works on a molecular level to swap unwanted minerals for harmless ones. This water treatment method is the industry standard for combating hardness because it physically removes the minerals rather than just preventing them from sticking to your pipes.
The Core Components: Mineral Tank, Brine Tank, and Control Valve
To truly understand how these systems operate, you need to look closer at the internal architecture hidden within these main components. A standard water softener relies on several highly specialized parts to maintain proper flow and chemical reactions:
- The Dip Tube: Often called a distributor tube, this long PVC pipe runs straight down the center of the mineral tank. When hard water enters the system, it flows down through the resin bed to be softened. The freshly softened water is then forced back up through the dip tube to be distributed throughout your home’s plumbing.
- Gravel Underbedding: Sitting at the very bottom of the mineral tank, a layer of specialized quartz gravel acts as a foundational support for the resin beads above it. It also helps evenly distribute the flow of water around the bottom distributor basket, preventing the system from channeling or losing vital water pressure.
- Brine Float Mechanism: Located inside the brine tank, this component functions much like the float in your toilet tank. It carefully controls the amount of water that enters the brine tank to mix with the salt, ensuring the perfect concentration of brine is created without overflowing onto your utility room floor.
The Science Simplified: How the Ion Exchange Process Works
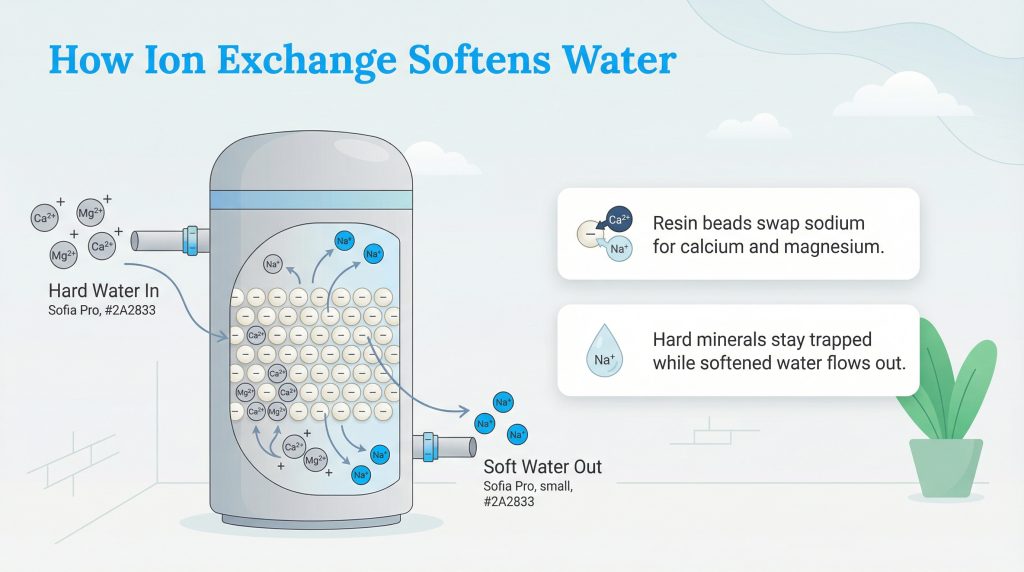
To understand how ion exchange water systems work, it helps to visualize a simple magnet. Inside the mineral tank of your water softener, there are thousands of tiny plastic spheres called ion exchange resin beads. These beads are negatively charged. Because opposites attract, they hold onto positively charged sodium ions. When hard water flows into the tank, it brings along calcium and magnesium minerals, which are known in chemistry as divalent ions (meaning they carry a +2 electrical charge, written as Ca2+ and Mg2+).
Because calcium and magnesium carry a double positive charge, they serve as much stronger magnets than the single-charge sodium ions (Na+). As the hard water passes over the resin bed, the beads instantly grab these divalent hard minerals. To maintain perfect electrical neutrality, the exact chemical exchange mechanism dictates that one hardness ion displaces exactly two sodium ions. The minerals that cause stubborn scale are effectively trapped on the beads, and the water that exits the tank contains only the harmless sodium ions, leaving it soft and safe for your household.
Polystyrene Resin Beads and Divalent Ions
The secret behind this highly efficient hard water treatment process lies in the physical structure of the beads themselves. Most top-tier water softeners use cross-linked polystyrene resin beads, which are synthesized to be extremely porous and durable. The chemical cross-linking process creates a microscopic, sponge-like polymer matrix. This porosity is absolutely critical because it vastly increases the surface area available for the chemical exchange to take place. As water flows through the myriad of internal pores, the divalent ions are forced into close contact with the exchange sites, ensuring virtually all hardness is trapped before the water ever reaches your faucets.
Cation vs. Anion Exchange: Understanding the Difference

While water softening is the most common use for this technology, the type of resin used depends entirely on what you need to remove from your water source. This brings us to the crucial difference between cation vs anion exchange resin. It all comes down to the fundamental electrical charge of the ions being targeted.
Cation Exchange resins are negatively charged and are used to attract positively charged ions. This is what you find in a standard water softener designed specifically for water softening. It targets calcium, magnesium, and sometimes iron. Occasionally, this is referred to as a deionization water system when used for highly complex, high-purity applications, though residential softeners specifically target hardness minerals. On the other hand, Anion Exchange resins are positively charged and attract negatively charged ions. These are typically used for removing nitrates, sulfates, or harmful arsenic from water supplies. Understanding which contaminants are present in your water ensures you select the precise resin type for your home’s unique needs.
The Regeneration Cycle: Keeping Your System Running
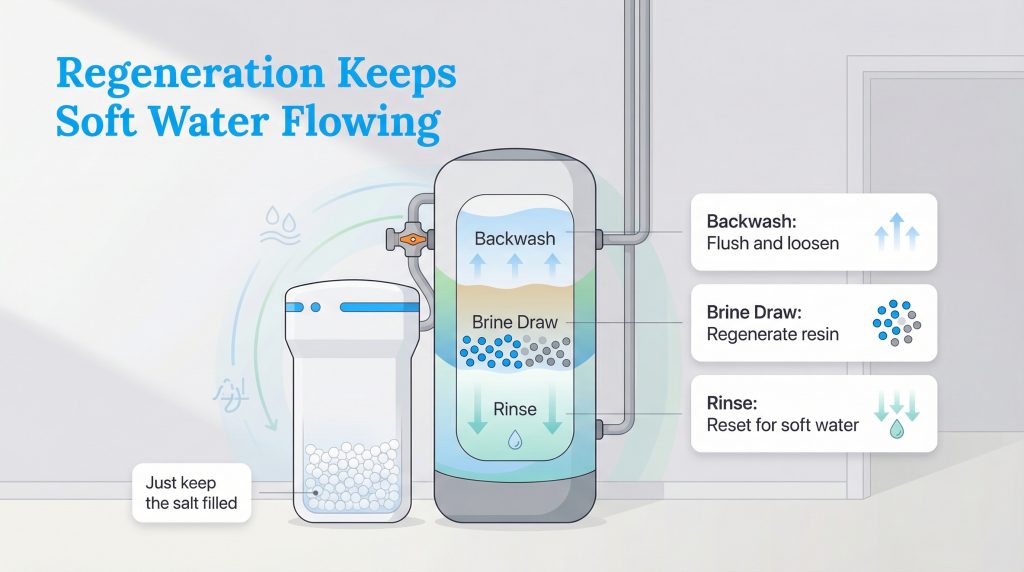
Eventually, those highly porous resin beads get “full” of hard water minerals and can no longer attract new ones. When this happens, the system must enter a water softener regeneration cycle to effectively clean itself. This automated process uses a remarkably strong salt solution from the brine tank to wash the beads.
During the ion exchange resin regeneration process, the control valve systematically floods the resin with brine. The sheer volume of sodium ions in the brine forces the stubborn calcium and magnesium off the beads and flushes them harmlessly down the drain. Here is exactly how the four distinct phases of the cycle flow:
The 4 Stages: Backwash, Brine Draw, Rinse, and Refill
- Backwash (10–15 Minutes): The control valve boldly reverses the flow of water, pushing it backward up through the mineral tank. This fast-flowing water actively expands the resin bed, aggressively flushing out accumulated dirt, sediment, and heavy iron particles down the drain. This crucial first step typically uses around 15 to 20 gallons of water.
- Brine Draw (50–60 Minutes): The system slowly pulls the highly concentrated, salty brine solution from the brine tank directly into the mineral tank. As this dense liquid cascades over the resin beads, the massive concentration of sodium overwhelms the trapped calcium and magnesium, breaking their magnetic bond. This long, slow phase uses about 10 to 15 gallons of water and successfully restores the positive charge to the beads.
- Rinse (10–20 Minutes): Once the hardness minerals are fully displaced, fresh water flows downward through the tank. The initial slow rinse pushes the remaining brine deep into the bed, while the subsequent fast rinse thoroughly washes away any leftover salt and settles the resin back into its tightly packed formation for daily service. Expect this to use 15 to 25 gallons of water.
- Refill (5–15 Minutes): Finally, the control valve sends a measured amount of fresh, softened water back into the brine tank. This step pre-dissolves a new batch of salt pellets so the strong brine solution is fully ready for the next scheduled regeneration cycle. It usually requires roughly 3 to 5 gallons, depending on the system’s size.
For you as the homeowner, this mechanical choreography is mostly automated. Your main job is simply to keep the brine tank filled with salt pellets so the system can reliably do its work in the background.
Sizing and Testing: Understanding Grains Per Gallon (GPG)

Before purchasing an ion exchange water system, you must determine the exact hardness level of your home’s water. In North America, water hardness is universally measured in Grains Per Gallon (GPG). One grain of hardness is equivalent to 17.1 milligrams of calcium or magnesium dissolved in one liter of water. Knowing your precise GPG is crucial because it dictates the overall size, or capacity, of the mineral tank you will need to buy.
| Water Classification | Grains Per Gallon (GPG) | Typical Home Impact |
|---|---|---|
| Soft | 0 – 1.0 GPG | No significant scale buildup; soap lathers perfectly. |
| Moderately Hard | 3.5 – 7.0 GPG | Minor spotting on dishes; appliances may require more maintenance. |
| Hard | 7.0 – 10.5 GPG | Noticeable scale on faucets; dry skin and higher water heating bills. |
| Very Hard | Over 10.5 GPG | Severe plumbing damage over time; requires a high-capacity softener. |
If you rely on municipal city water, you can often check your local water utility’s annual consumer confidence report for an average hardness rating. However, public water quality fluctuates, so testing it at your actual tap is always the most accurate method. Here is how to use simple hardness test strips to calculate your required tank capacity:
- Dip the Test Strip: Fill a clean glass with cold tap water. Briefly dip a standard water hardness test strip into the water for about one second, then pull it out and gently shake off the excess. Wait roughly 15 seconds for the color pad to fully react.
- Read the Results: Compare the color on the strip to the provided chart on the testing kit’s bottle. This will give you your water hardness in GPG. (If the chart uses parts per million, simply divide that number by 17.1 to get your GPG).
- Calculate Your Daily Requirement: Multiply your GPG by your household’s estimated daily water usage. The industry average is roughly 75 gallons per person, per day. For example, a family of four uses about 300 gallons daily. If your water is 10 GPG, your system must systematically remove 3,000 grains of hardness every single day (300 gallons x 10 GPG). This calculation allows you to buy a softener with the correct grain capacity — such as a 32,000-grain or 48,000-grain unit — so it doesn’t have to regenerate constantly.
The Environmental and Health Impact of Ion Exchange Systems

While exceptionally effective, ion exchange systems do have an environmental and health footprint, primarily regarding water usage and sodium discharge. A common concern is how much sodium the process actually adds to household drinking water. The chemistry is exact: approximately 8 milligrams per liter (mg/L) of sodium is added to the water for every grain of hardness removed. If your water is 10 GPG, you are adding about 80 mg/L of sodium to your tap water, which is a very mild amount for most adults but something to consider if you are on a strict, medically supervised low-sodium diet.
On the environmental side, older systems operated on a strict timer, regenerating every few days regardless of actual water use, which needlessly wasted both salt and water. Modern systems use Demand-Initiated Regeneration (DIR), which actively monitors your water usage through a digital meter and only regenerates when absolutely necessary. This is a far more water efficiency focused approach that saves money and precious ecological resources.
For those deeply concerned about salt, there are salt-free conditioners available. However, it is important to note that these are not true softeners; they don’t remove hardness minerals but rather change how the minerals behave so they are less likely to stick to surfaces. If you want the removal benefits of ion exchange but want an eco-conscious alternative to standard salt, you have options. We also strongly recommend checking your local municipal codes, as some areas have strict restrictions on salt-based softeners due to local wastewater concerns.
Is an Ion Exchange System Right for Your Home?
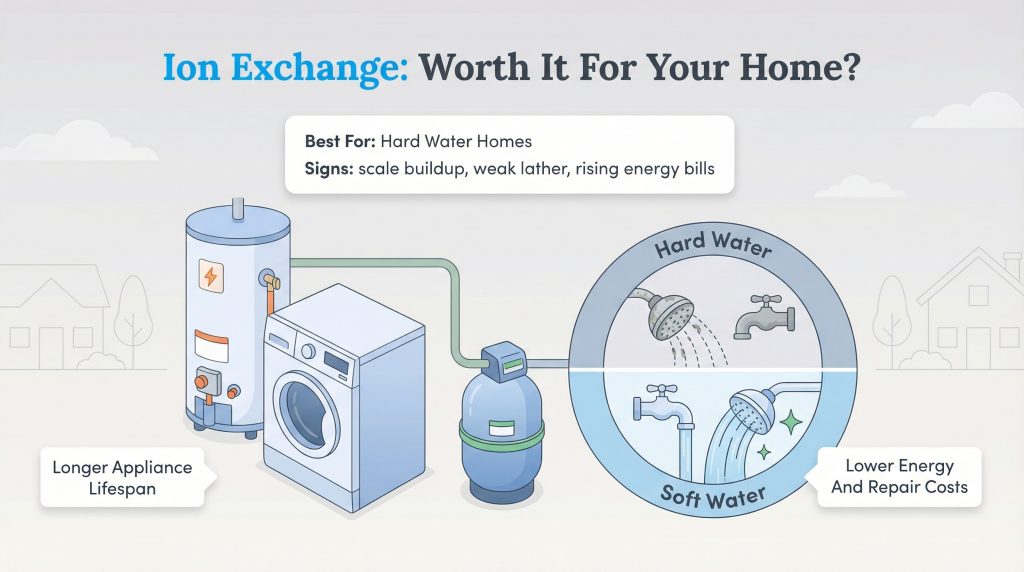
When you are deciding whether to install a system, you will balance the advantages of ion exchange water treatment against the upfront cost. If your water hardness is above 7 GPG, you likely notice crusty scale on showerheads, poor soap lather, and creeping water heating bills. Transitioning from hard water to soft water ion exchange can easily extend the lifespan of your washing machine, dishwasher, and water heater by years. Weigh the pros and cons carefully before committing:
| Pros | Trade-Offs |
|---|---|
| Extends the life of appliances | Requires regular salt purchases |
| Reduces energy costs for water heating | Discharges brine into wastewater |
| Cleaner dishes and softer laundry | Slight increase in sodium content |
While the initial investment includes the unit and professional installation, the impressive long-term savings on appliance repairs and energy efficiency often pay for the system over time. You can learn more about how efficient appliances impact your utility bills in our guide on how to save on your electric bill. You can also explore more about how general water quality affects your monthly utility costs in our comprehensive guide to residential water services.
The Verdict on Upgrading Your Home’s Water Quality
Ion exchange is a proven, thoroughly reliable way to banish hard water headaches from your home. By aggressively swapping troublesome, scale-forming calcium and magnesium for harmless sodium, these systems protect the hidden infrastructure of your property. The long-term return on investment becomes incredibly clear when you factor in the extended lifespan of your water heater, washing machine, and dishwasher. Scale buildup acts as a stubborn insulator on internal heating elements, forcing your appliances to work much harder and consume significantly more electricity or natural gas. By investing in a well-sized water softener, you are actively safeguarding your home’s plumbing health and ensuring your most expensive appliances run efficiently for years longer than they ever would on hard water.
Whether you choose a standard salt-based system or opt for potassium chloride, understanding the internal regeneration cycle and basic component maintenance ensures your water stays perfectly soft. We highly encourage you to purchase a hardness test strip today to accurately measure your home’s GPG. If your readings come back exceptionally high, or if you feel overwhelmed by the technical sizing calculations, reach out to a local water utility specialist to explore professional installation options. Taking action now is the first definitive step toward a cleaner, more energy-efficient home.
Learn more about Water Filtration for your Home.
FAQs About Ion Exchange Systems
What is the difference between ion exchange and reverse osmosis?
How long does ion exchange resin last?
Does ion exchange remove bacteria?
Is ion exchange water safe to drink?
Can I install an ion exchange system myself?
How much sodium does an ion exchange water softener add to my drinking water?
What is the difference between water demineralization and deionization?
Can I use potassium chloride instead of sodium chloride in my brine tank?
About the Author
LaLeesha has a Masters degree in English and enjoys writing whenever she has the chance. She is passionate about gardening, reducing her carbon footprint, and protecting the environment.
